A modified Solvay process with low‐temperature calcination of NaHCO3 using monoethanolamine: Solubility determination and thermodynamic modeling - Wang - 2019 - AIChE Journal - Wiley Online Library

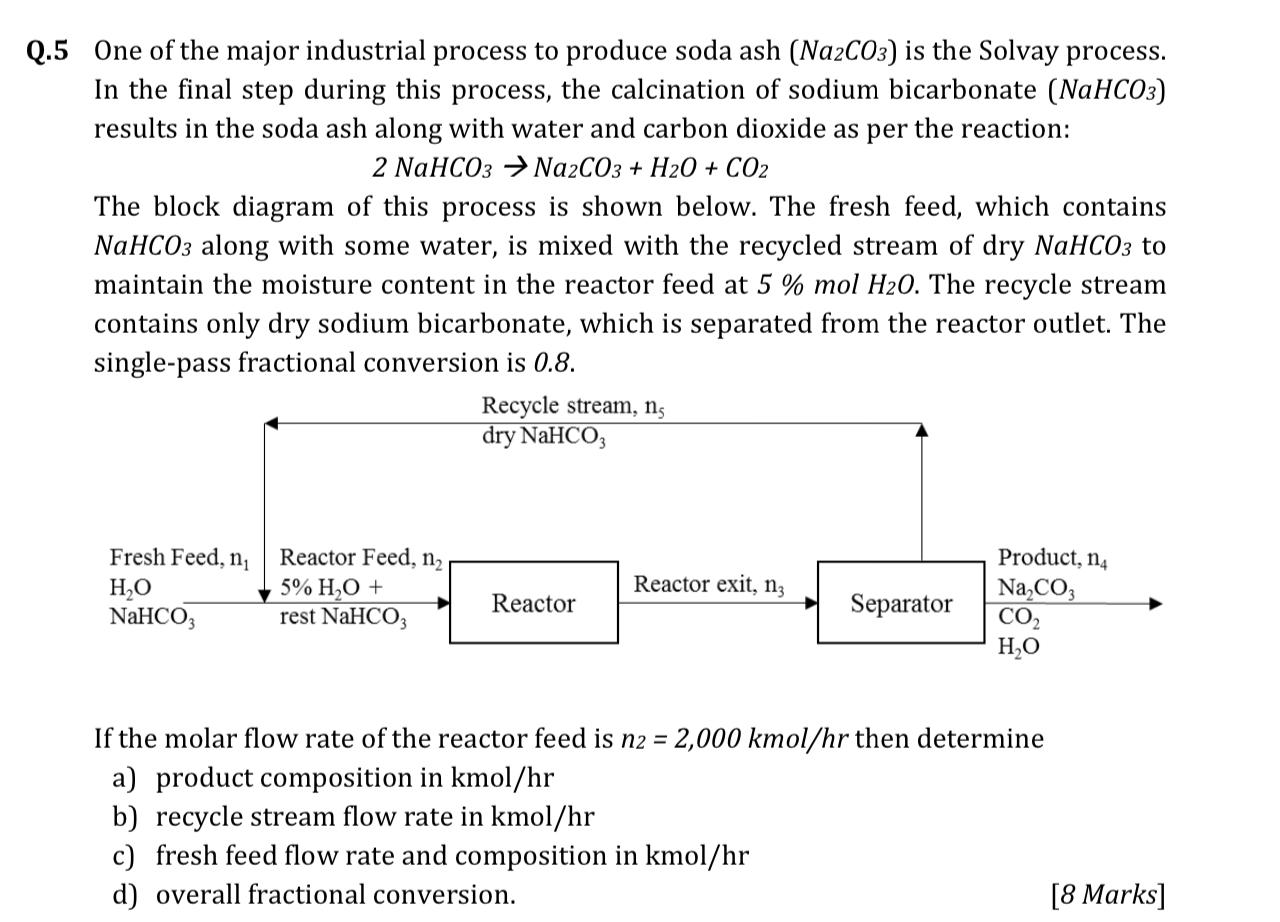
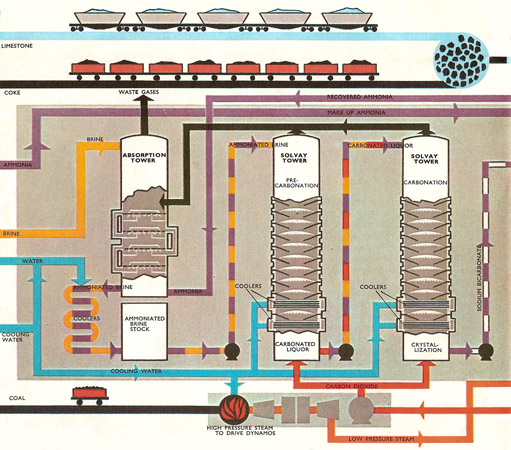
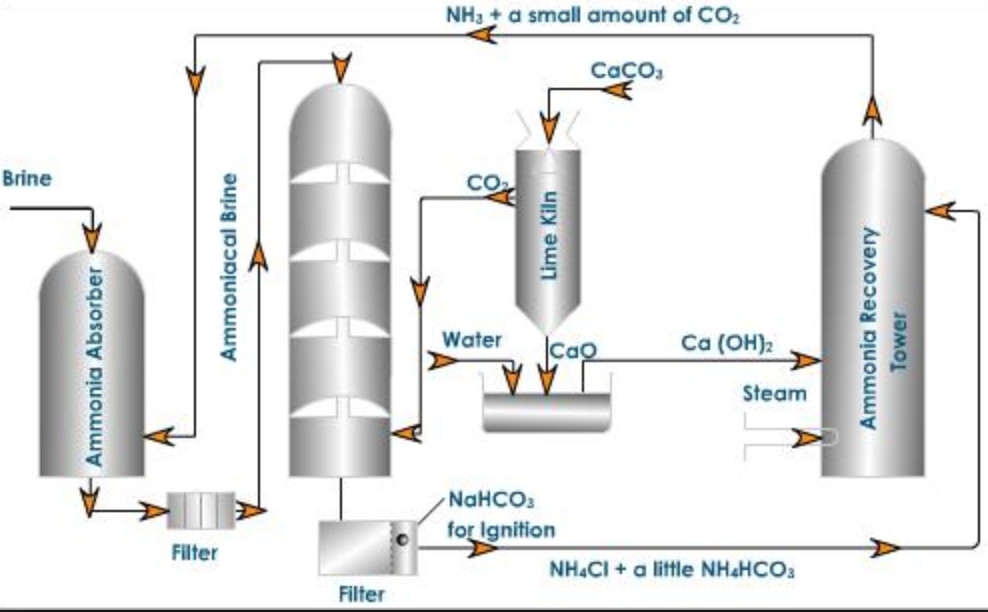
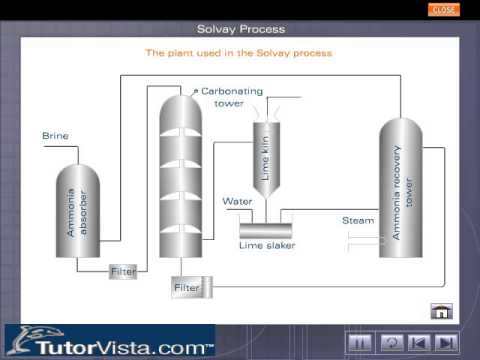
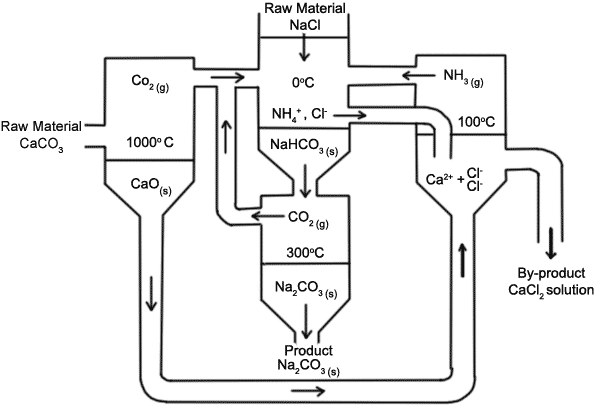
Chemical Technology – Solvay Process- Process Description-PFD-Chemical Kinetics-Engineering Problems – Chemical Engineering Facts
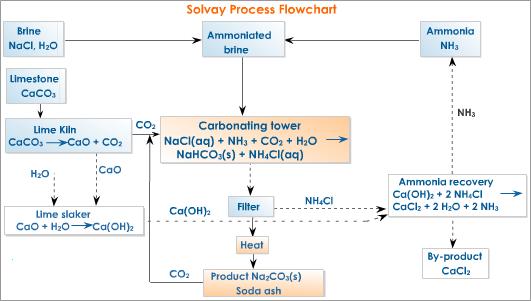
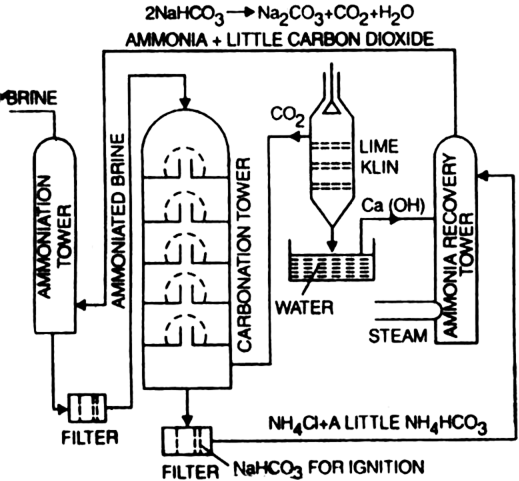
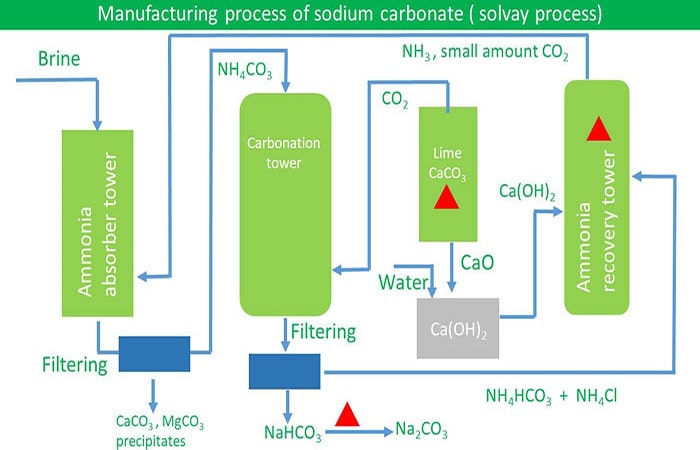
Describe Solvay process (or) how is washing soda (or) sodium carbonate prepared in industries? - Sarthaks eConnect | Largest Online Education Community

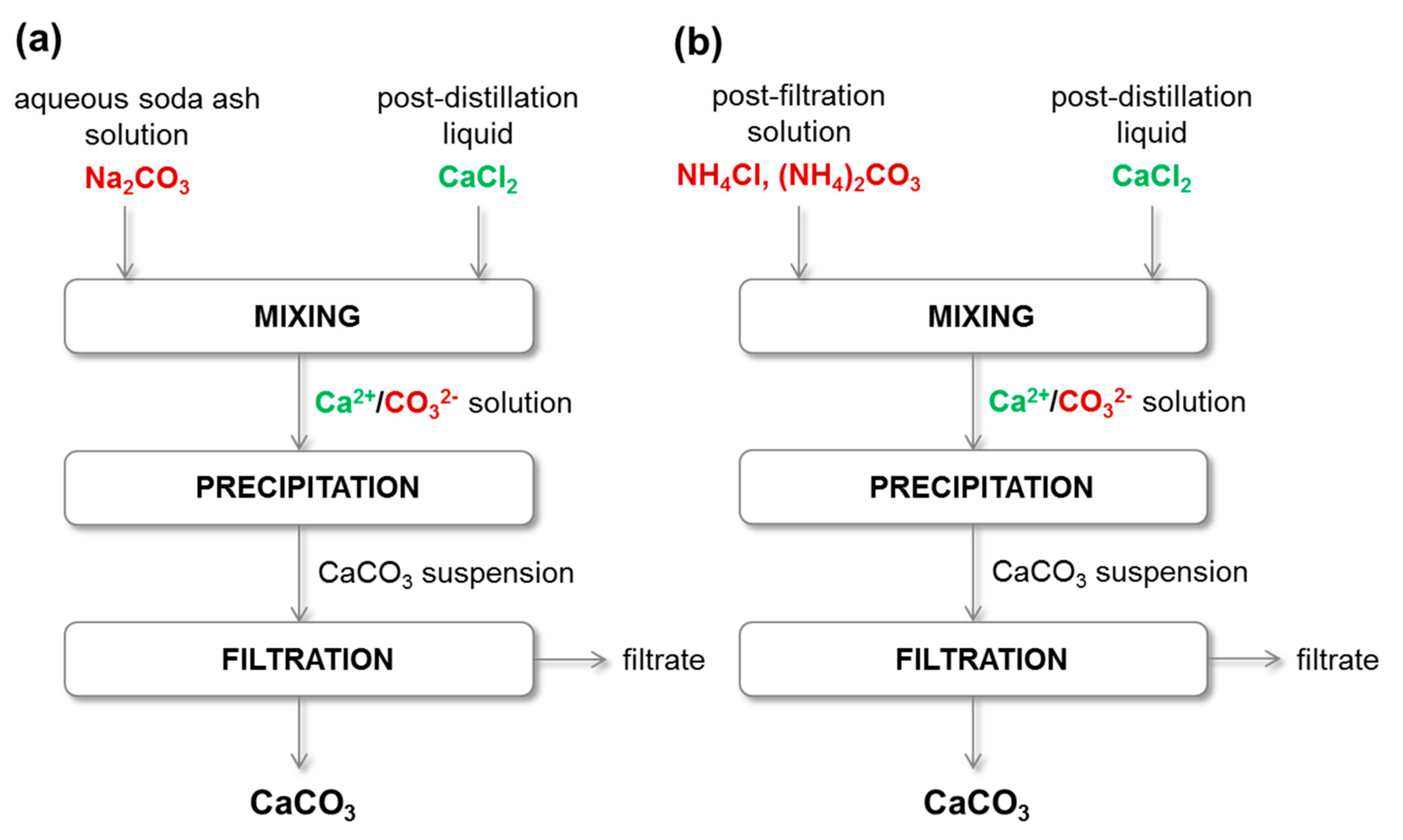
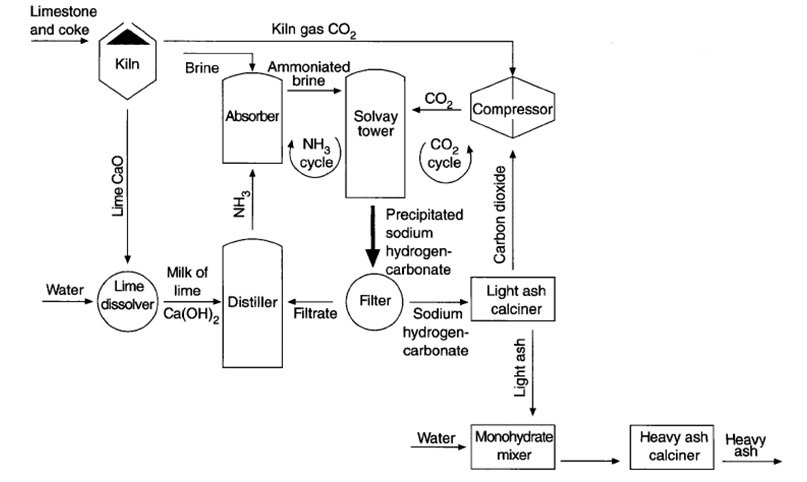
Figure 7 | A cleaner production of sodium hydrogen carbonate: partial replacement of lime by steel slag milk in the ammonia recovery step of the Solvay process | SpringerLink